Pharmaceutical
Coding pharmaceutical products
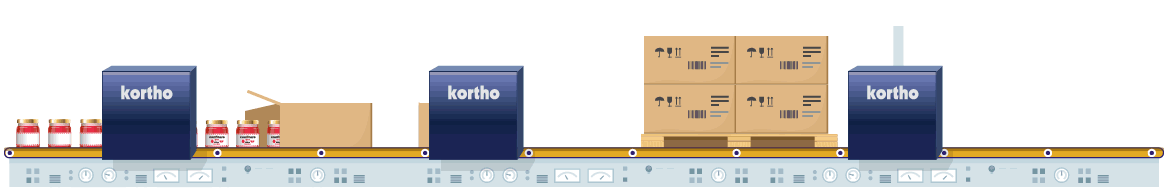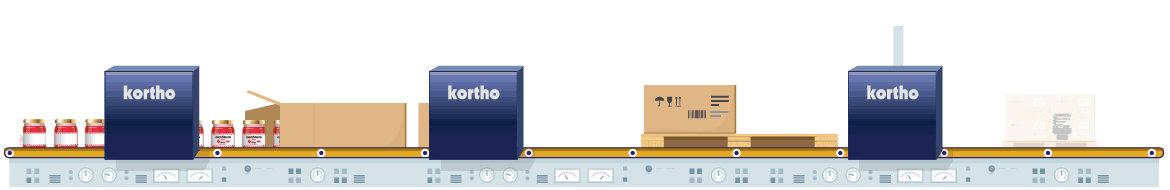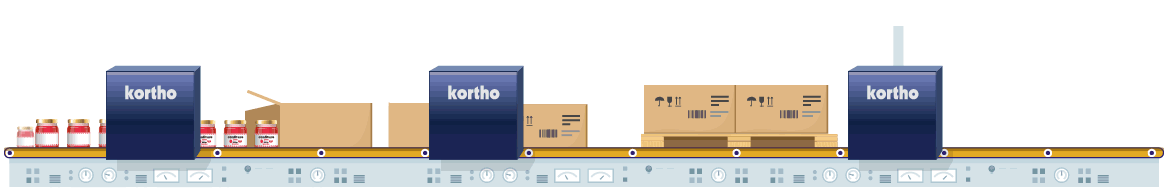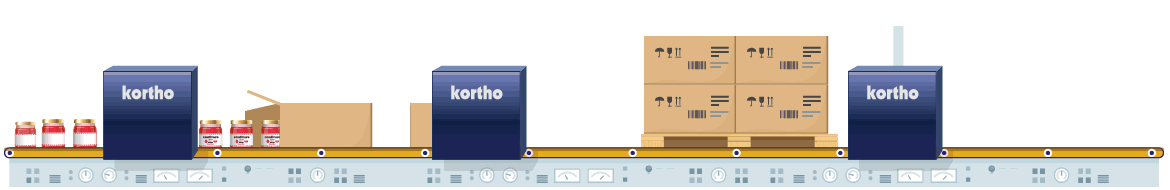
The manufacturing of pharmaceutical products is strictly regulated by law. Coding is a crucial aspect of the process. Only foolproof traceability and flawless product composition information, plus accurate expiry dates can guarantee safe product consumption. The fight against counterfeit products also influences this need. Conforming to 100% error-free coding regulations and the use of anti-counterfeit techniques are therefore critical in order to comply with relevant legislation and to eliminate opportunities for counterfeit imitation products.
Industry focused solutions for coding pharmaceutical products
Coding errors definitely need to be eliminated from individual product to pallet. Reliable equipment with direct control via the ERP is therefore essential. By conducting additional physical coding checks afterwards, any errors in the coding process will be eliminated from start to finish. Techniques such as serialisation also prevent the risk of counterfeiting. This process provides every product with its own unique code which is logged. Comparison of logged data to product codes at a later stage will validate authenticity of the products.